It has a low density but high strength and is resistant to both seawater and chlorine. Discovered in Cornwall in 1791 by William Gregor, titanium is an extremely useful transition metal. One further reaction involving group 2 metals uses magnesium to extract titanium (Ti). With a salinity of over 30 percent, this sea is one of the world's saltiest bodies of water. Magnesium chloride (MgCl 2) is the primary salt found in the Dead Sea. The chloride ions are spectator ions they are neither oxidised nor reduced, but rather keep the same physical and oxidation state. $$M+2HCl(aq)\rightarrow MCl_2(aq)+H_2(g)$$Īs before, the metal is oxidised, whilst hydrogen is reduced. This is the simplest case - group 2 elements react with hydrochloric acid (HCl) to give a metal chloride (MCl 2)and hydrogen gas (H 2): The reactions between group 2 metals and acids vary depending on the type of acid used, but all form colourless solutions of ionic salts. Group 2 reactivity with acidsįinally, we'll turn our attention to the reactions of group 2 with acids. The reactivity between group 2 elements and chlorine increases as you go down the group, thanks to the decreasing ionisation energy of the group 2 metal. In fact, group 2 metals react similarly with all halides. Group 2 metals react with chlorine (Cl 2) to produce metal chlorides (MCl 2). Next up: the reaction between group 2 elements and chlorine. Their reactivity depends on many factors, such as the heat needed to start the reaction and whether the metal is covered with a protective layer of metal oxide or not. 
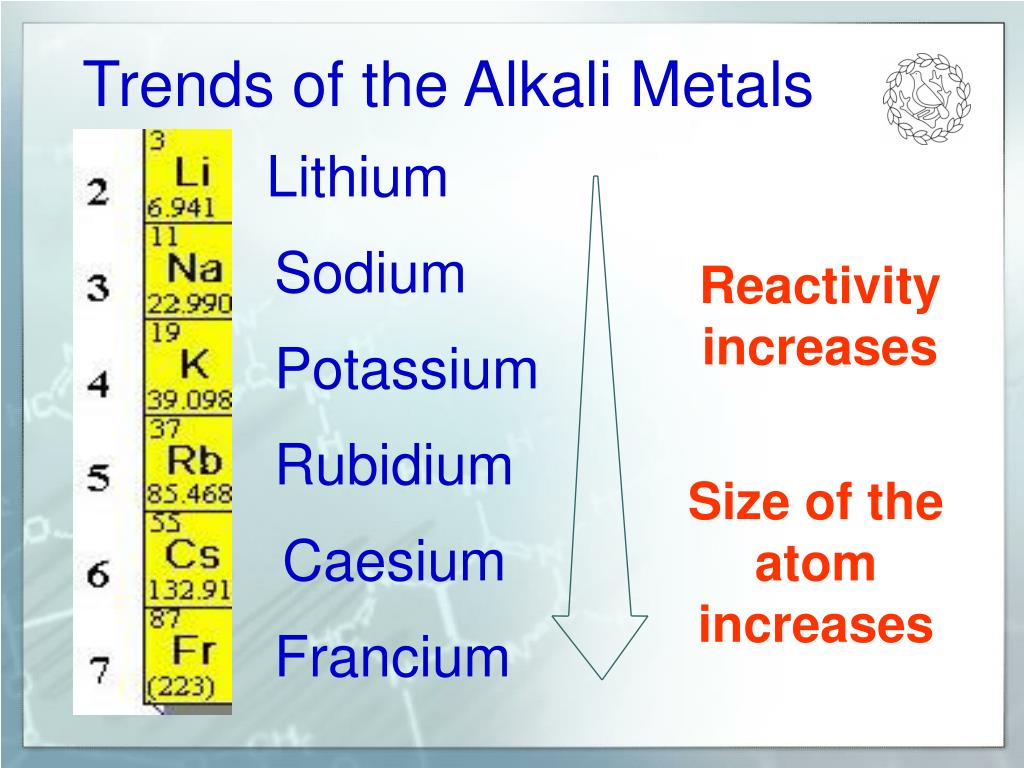
It is hard to give clear trends when it comes to the reactivity of group 2 metals with oxygen. The reaction initially forms magnesium hydroxide (Mg(OH) 2), but this species then splits up upon heating to produce magnesium oxide (MgO) and hydrogen gas (H 2). Magnesium also reacts with steam to produce the same product, magnesium oxide. We'll finish by summarising all that we've learned in a handy table before.We'll also find out about a further reaction of magnesium, used to extract titanium industrially.This will involve their reactions with water (to produce hydroxides), oxygen, chlorine, and acids.We'll then dive into the redox reaction of group 2.We'll define group 2 and briefly look at their electron configurations.This article is about group 2 reactivity in inorganic chemistry.In this article, we'll explore these reactions and look more closely at group 2 reactivity. This is because they always react with other species to form ions and compounds. But we never find magnesium, or in fact any of the other group 2 metals, in their elemental states in nature. Did you know that magnesium, a group 2 metal, is the fourth most common element found in the Earth itself, and the third most common ion in seawater? It is essential to human life too magnesium plays a role in the structures of 300 different enzymes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
